Reagent and supplies
| Reagent | Source | Catalog Number |
|---|---|---|
| CARD HyperOva FD | Cosmo Bio Co | KYD-015-EX-X5 |
| hCG 5000 IU | Sigma | CG5-1VL |
| 1 ml syringes micro-fine insulin | Fisher | 14-829-1B |
| CARD media kit (CARD medium + FERTIUP) | Cosmo Bio Co | KYD-008-02-EX-X5 |
| EmbryoMax KSOM Advanced | Sigma | MR-101-D |
| EmbryoMax mineral oil | Sigma | ES-005-C |
| Pipetman 20 ul, 200 ul, 1000 ul | Rainin | P-START |
| Pipette tips | VWR | 470335-046, 470335-048, 470335-050 |
| Wide bore pipette tips | VWR | 76635-646 |
| Plastic bulb transfer pipets | Fisher | 13-711-9CM |
| 60 mm culture dishes | Fisher | 0877221 |
| Long forceps | Fisher | 10316C |
| Dissection instruments (medium scissors, fine scissors, fine #5 forceps, serrated fine forceps) | Fine Science Tools | 14002-12, 14060-10, 11250-20, 11150-10 |
| Glass capillaries for embryo handling | Fisher | 13-678-20D |
| 3 to 5 week old female mice (weanling/pre-pubertal) |
IVF Procedure (using cryopreserved sperm in straws)
- Day 1 — Superovulation
- Superovulate 3 to 5 week old female mice
- Inject 100 μl CARD HyperOva intraperitoneally (i.p.) between 2:00 pm and 6:00 pm.
Day 3 — hCG and preparation
- 48 to 52 hours after HyperOva injection, administer 5 IU hCG i.p.
- Place beaker of distilled water in the incubator to warm overnight (for sperm thawing).
- Day 4 — IVF Day
- Prepare CARD fertilization medium (formulation depends on sperm type).
- Prepare mHTF washing medium or KSOM Advanced (MR-101D, Sigma) if using alternative.
- Prepare culture dishes:
- Sperm dish
- One 60 mm dish per sperm straw/vial.
- Add 90 μl of FERTIUP sperm preincubation medium.
- Cover with mineral oil.
- Place in the incubator 30 minutes before thawing sperm.
- Fertilization dish
- One 60 mm dish per superovulated injected female.
- Add 200 μl CARD fertilization medium for fresh sperm or 90 μl CARD fertilization medium for frozen sperm.
- Cover with mineral oil.
- Place in the incubator 10 minutes before sacrificing the first female.
- Washing dishes
- One 60 mm dish per fertilization dish.
- Add four 80 μl drops of mHTF or KSOM, cover with mineral oil.
- Place in the incubator at least 30 minutes before embryo rinsing. (These can also be prepared immediately after insemination.)
- Sperm dish
Collection of oocytes:
- Sacrifice each female 15 to 17 hours after hCG. Process one female at a time.
- Expose the ovaries/oviducts and remove oviducts, minimizing fat, blood, and fluid.
- Place oviducts into the oil layer of the fertilization dish.
- Tear open the ampulla with #5 forceps to release the cumulus-oocyte complexes (COC).
- Drag the COCs into the CARD medium drop and return dish to the incubator.
- After all oocytes are collected, incubate fertilization dishes for 30 to 60 minutes before insemination.
Thaw sperm:
- Remove a frozen sperm straw from liquid nitrogen; hold in air for 5 seconds.
- Immerse straw in 37°C water for 10 minutes.
- Remove straw, wipe with 70% ethanol.
- Cut off the seal and cut through the PVA plug, leaving half of the cotton plug.
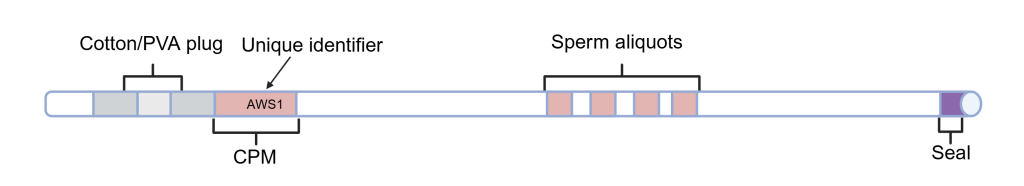 (Created in https://BioRender.com)
(Created in https://BioRender.com) - Using a metal rod as a plunger, push against the remaining portion of the cotton plug and expel the contents into the FERTIUP drop; avoid dropping the plug.
- Incubate sperm dish for 30 minutes.
Insemination:
- Pipette 10 μl of sperm suspension from the edge of the FERTIUP drop (most fertile sperm).
- Add sperm directly onto the cumulus masses in the fertilization dishes.
- Return dishes to the incubator.
Post-Insemination Handling:
3 hours post-insemination:
- Wash oocytes three times through the mHTF or KSOM drops.
- Leave oocytes in the third mHTF or KSOM drops overnight.
6 hours post-insemination:
- Examine oocytes and discard unfertilized ones.
- Continue overnight incubation.
Day 5 – Embryo Handling
- The next morning, transfer two-cell embryos to the fourth mHTF drop.
- Embryos may now be cryopreserved, cultured, or transferred to 0.5 dpc recipients.
Variations in IVF Procedure Depending on Sperm Source
Fresh Sperm
- Sacrifice the male and remove the cauda epididymides, minimizing fat, blood, and excess tissue fluid.
- Place the cauda epididymides into the oil layer surrounding the FERTIUP sperm drop.
- Using small sharp scissors, cut the duct.
- Gently press the cauda surface to release sperm.
- With sharp forceps, drag sperm clots into the FERTIUP drop.
Capacitation
- Incubate the sperm suspension for 60 minutes to allow capacitation.
Insemination
- Inseminate COCs with 3 μl of the sperm suspension.
- Proceed to the Post-Insemination Handling of the IVF CARD protocol.
Frozen Sperm in Vials
Follow Steps 1-5 of the IVF CARD protocol, then:
- Remove a frozen sperm vial from liquid nitrogen. Open the cap carefully and discard any LN2 inside the tube.
- Place the vial in a 37°C water bath for 10 minutes.
- Transfer the thawed sperm suspension to a 1.5 mL tube using a wide‑bore pipette tip.
- Slowly add 1.2 mL warmed mHTF, then centrifuge at 300 g for 5 minutes at room temperature.
- Carefully remove as much supernatant as possible.
- Add 70 µL warmed FERTIUP to the pellet (final volume ~100 µL).
- Transfer the entire sperm suspension into the 100 µL FERTIUP drop (sperm dish) using a wide‑bore tip.
- Incubate the sperm dish for 30 minutes.
Insemination
- Using a wide‑bore pipette tip, collect pre‑incubated COCs with minimal CARD medium.
- Release COCs into the sperm suspension drop.
- After 3 hours, proceed to the Post-Insemination Handling of the IVF CARD protocol.
Alternative Method for Thawed Sperm (Recommended Variation)
(As described in the CARD book, Cosmo Bio)
- Thaw sperm exactly as described in the frozen‑vial method above.
- Pipet the thawed sperm directly in the pre‑incubated FERTIUP drop.
- After the standard 30‑minute incubation, proceed with insemination as described for fresh sperm.
Notes and Critical Considerations
Media Preparation
- CARD medium formulation depends on sperm source. Follow manufacturer instructions precisely.
Handling of Females
- Sacrifice females one at a time.
- Work quickly to transfer COCs into CARD medium and return dishes to the incubator promptly.
Sperm Handling
- Do not disturb sperm dishes during pre‑incubation. Movement can prevent sperm from achieving full motility.
- For cryopreserved sperm vials – After thawing, perform a quick visual assessment of a small aliquot of sperm. Based on this evaluation, divide the thawed vial appropriately among the prepared FERTIUP drops. Typically, three FERTIUP drops are set up for pre‑incubation to ensure sufficient options if needed.